[New Product Release] The standard for malignant tumor-27 gene-38 sites of the blood system, come and apply for a trial installation ~
We have launched the standard of vicious tumor of hematopoietic system, involving bone marrow hyperplasia abnormal syndrome (MDS)/bone marrow proliferative tumor (MPN)/acute marrow leukemia (AML)/chronic granulocyte leukemia (CML)/chronic lymphocyte leukemia ( CLL)/acute lymphocytic leukemia (ALL) and other common genetic mutation points related to diseases.
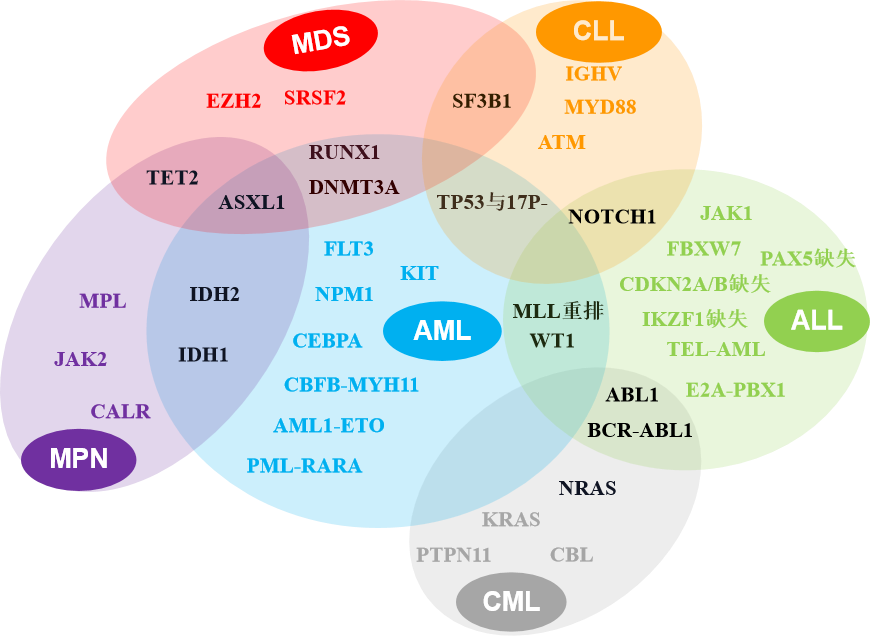
Fig 1. Common blood system malignant tumor classification and related genetic sites
In order to thank you for your trust and support for us you have launched the "New Product Trial" activity. 10 places, first come first, first, welcome to actively apply for a trial dress. For the application method, see the end of the article.
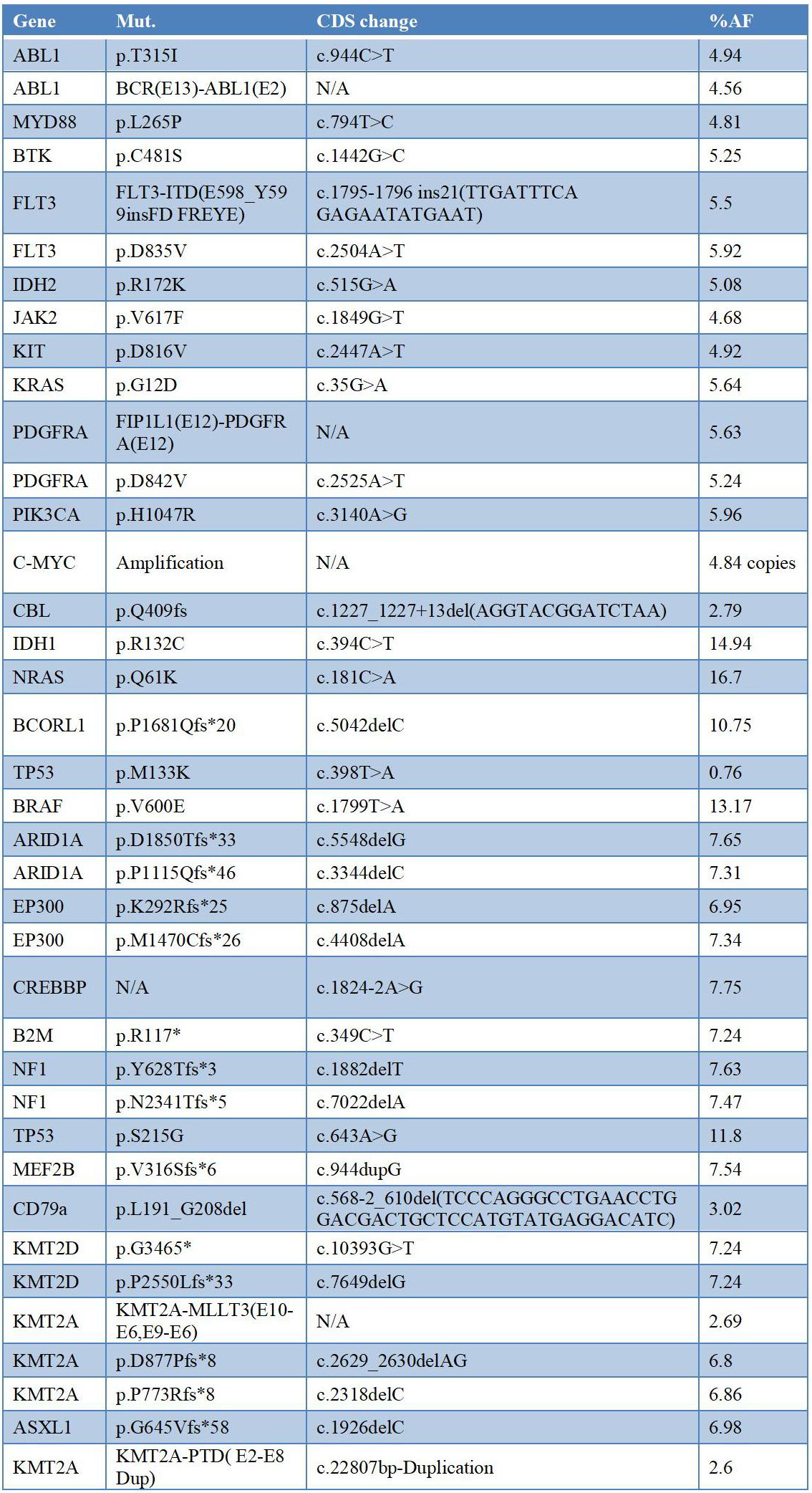
This is a multi -gene and multi -digit Panel standard product. The raw materials used in mixed samples are basically derived from the blood system -related tumor samples. Kai Bai Bio has 38 bit -related points related to 27 genes, all of which have developed a digital PCR (DDPCR) detection system, and accurately standardized the mutant frequency or copy number variation value corresponding to the benchmark. And most of the mutations are pathological significance.
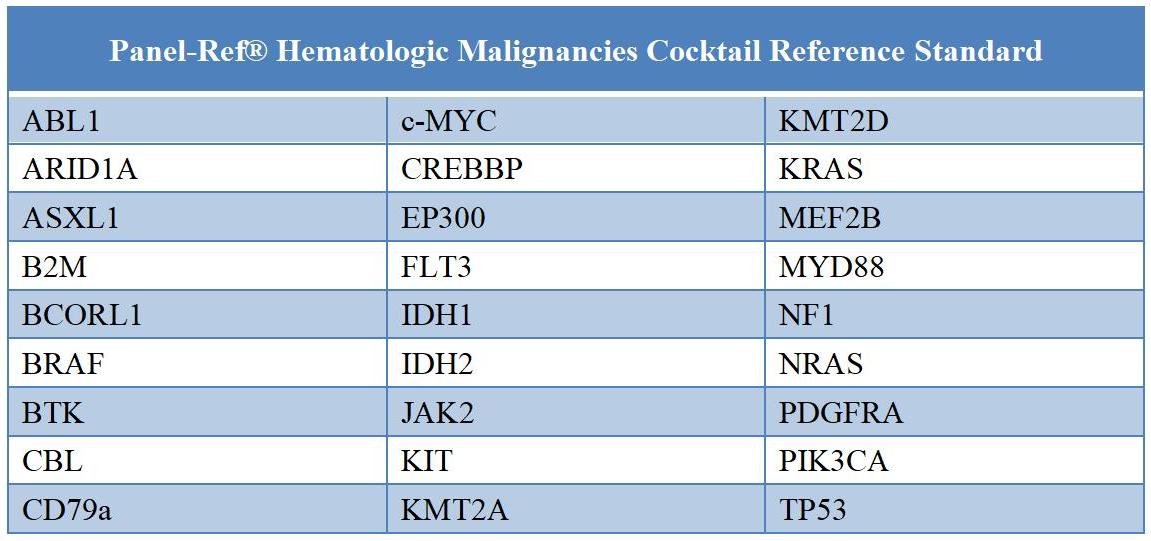
Table 1. 27 genes related to Panel related to the blood system malignant tumor Panel
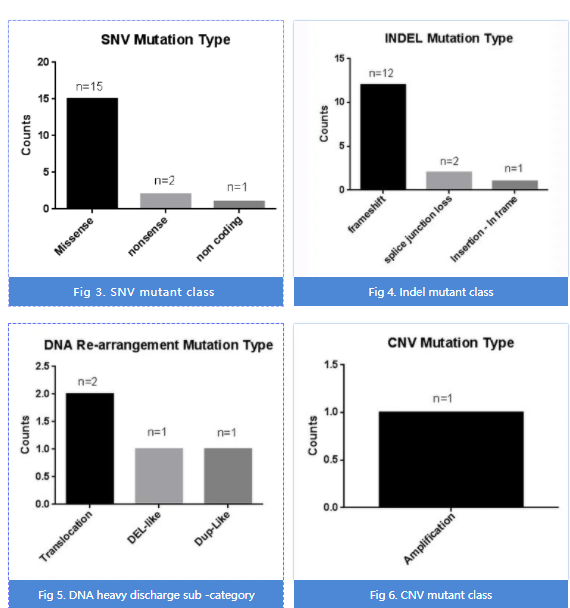
Most of the insertions and lack of duplicate bases are repeated), 4 DNAs are rearranged, and 1 copy number mutation.
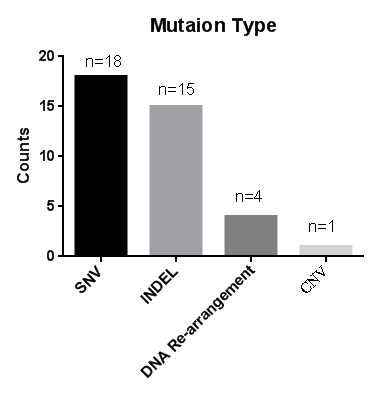
Fig 2. 38个位点对应的突变类型
The mutation frequency is around 5%, and it can be used for further dilution, as a LOD reference product, MRD reference product, etc.
The corresponding clinical case grading is mainly Uncertain Significance, Likely Pathogenic, Pathogenic, and according to CLINVAR annotations (some other databases that have not been noted by Clinvar), including 12 UNCERTAIN SIGNIFICANCANCE, 3 Likely Pathogenic and 3 Likely Pathogenic and 23 Pathogenic.
Panel-Ref® Hematologic Malignancies Cocktail Reference Standard RQP90039